HTF SF Recap: Digital Health in Pharma (9/27/17)
In this post, I highlight key takeaways from the latest HTF San Francisco event.
Background
The U.S. and New Zealand are the only two developed countries that allow direct to consumer drug advertising.
The American drug market accounts for half of the global pharmaceutical market, and 2016 pharmaceutical sales in the U.S. was estimated to be $446 billion dollars (for some perspective, the global coffee industry is just over $100 billion).
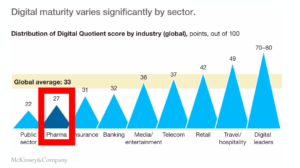
But despite being one of the biggest industries in America, pharma has the lowest digital maturity of any private sector. There’s much more they can do.
And with Amazon considering making a move to sell drugs online, the digitization of pharma isn’t far away.
As the U.S. continues to move toward a value-based model, thepharma industry will have to figure out how to use new technology to increase adherence, reduce drug development costs and time to market, improve and prove patient efficacy, lower prices and transition to a new business model.
Speaker #1
Jessica Chao, PharmD, MBA is the Director for the Clinical Innovation Center (CIC) at UCSF where she oversees day-to-day operations and leads CIC partnerships with industry and academia. Previously, she led product design in various San Francisco startups and co-founded Healtho, a consumer health information company based in Seattle. She is also the co-founder of one of the largest health Meetups in Seattle.
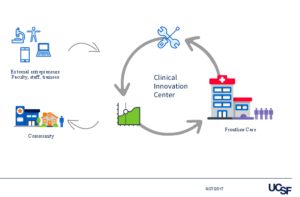
The CIC is a new group at UCSF with close ties to its hospital operations. Now in its second operating year, the center aims to incubate digital health solutions and integrate them into the hospital workflows and care pathways. CIC’s mission is to accelerate innovation for care delivery at UCSF.
The CIC also partners with external groups (startups, pharma, academia, data scientists) if they need additional expertise on a project to get to targets as soon as possible. The solutions that come out can be commercialized and brought back to the community.
Digital Health Solutions and Complex Patient Population
The main focus of Jessica’s group is to tackle continuation of care and see how digital solutions fit into pathways within the hospital.
When working on a digital health solution, it’s important to take into consideration the existing care pathways. The complexities of healthcare lie in the intersections of workflows, and that is also where the opportunities are for change, innovation and transformation.
Translating digital solutions and evidence-based medicine into care practice is a process that involves many moving parts.
Since 25% of patients in the hospital experience some type of delirium (up to 70-90% in the ICU), the CIC implemented a new care delivery pathway in the neurology unit to explore the use of an active delirium monitoring tool. This required coordination with many different members of the care team across the organization, and various EMR modifications. This is typical, as any new care model or digital solution introduced affect many different professions within the hospital ecosystem.
In order to implement and communicate a new pathway, it takes a large scale effort. And even when it is successful, digital solutions usually only tackle a part of the problem due to its complexity. An interdisciplinary approach with multiple integrated digital solutions are needed to address root causes in complex patient conditions.
Management of High Cost and High Risk Pharmaceuticals
UCSF prescribes emerging therapies as a research institution, which are expensive due to the companion labs, additional diagnostics and monitoring required in order to ensure patients are being prescribed therapies that are making them better.
For the health system, it’s not just the drug but the continuum of care needed for the patient that’s driving up costs. Digital solutions have huge potentials to manage increasing costs of pharmaceuticals.
Different Roles Digital Technologies Can Play:
- Ability to monitor different cohorts
- Provide ongoing education for different types of patient touchpoints
- Make decision for patient safely and take ongoing cost of management into account along with efficacy
- Robotics to make drugs faster, more efficiently and manage inventory
Speaker #2
Wayne Pan, MD, PhD, MBA, has more than 20 years of executive healthcare experience spanning managed care, health information technology, biotechnology and clinical practice as a orthopaedic hand surgeon. He talks about where pharma is today and where pharma should be tomorrow and unveils the story of mPharma (a tribute to the Tony Award-Winning play M. Butterfly).
The Story of Pharma
This story has three main characters: Science, Patients and Financing. These are the driving forces of the business model, and the underlying theme is tradition versus transformation. The pharma tradition lies in drug discovery, development and heavy marketing.
The transformation lies in what digital health has the potential to do, such as identifying biomarkers related to disease states personalized healthcare, and value-based purchasing.
Healthcare consumerism and personalized healthcare is on the rise and driving the transformation, as consumers don’t pay for things that aren’t tailored towards them. For example, mental health drugs don’t work for 50% of the patients they’re prescribed for. That isn’t acceptable in the dynamics of a consumer-driven marketplace, and there will be expectations of drugs that match both the disease and personal biology.
Pharma is the Least Digitally Mature Private Sector
5 Factors Keeping Pharma traditions in Place:
- Conservative Culture: high regulated industry, very cautious about making changes
- Compliance: don’t want to raise any FDA/FTC flags
- Global Organization: typically large international companies. Must adhere to all drug agency standards all over the world, not just in the US
- Long-term investments: drug discovery and development process takes billions of dollars per drug and are multiple year projects.
- Inertia: the larger the organization, the harder it is to get it to move in a new direction
5 Factors Pushing for Change and Transformation:
- Competition (others starting to change)
- Business model shifting (value-based care)
- Sustainability (pivoting to remain relevant)
- Innovation (emerging technologies)
- Agility (get into different markets and not remain rigid)
There are 3 areas where digital health can improve the Pharma Experience: Understanding the patient, expediting the science and enabling the financing.
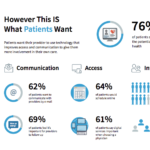
Right now, patient understanding comes only through lab tests, charts and clinical trials. The system only engages when patient is in front of the physician.
Behavior: what they don’t know about is the 99% of time when patients are not in front of a doctor, how do patients respond to drugs in the real world, how do their diseases manifest themselves outside of the clinic environment, are there subtle signs to inform when a disease state changes Right now patients may be asked to journal their symptoms manually, but this data can be difficult both for patients to report and for physicians to incorporate into their workflow and decision-making process (are treatments working, what should be changed). Digital technology solutions that can automatically collect patient reported data and summarize it when the patient returns for a visit either for clinical trial or doctor appointment might be extremely helpful. Valuable to help determine if therapy is working.
Genome & Proteome: Genomic and proteomic information to support clinical decision-making can streamline treatment decisions but doesn’t replace clinical interpretation and human interaction. We know that health outcomes are more than the sum of your DNA and expressed protein mix.
Biomarkers: Just as we will have better understanding of the genetic and proteomic make up of people, we are progressing towards a better understanding of how drugs interact with people and are developing diagnostic capability to determine through biomarkers, whether a particular therapy will be effective for patients who express a particular biomarker(s).
Home monitoring: pharma isn’t exploiting this realm right now, but being able to collect data on patient activities in the home setting can be an important piece of information needed by clinicians in assessing how patients are doing – either in a regular clinical setting or in a clinical trial. Obtaining this information now is difficult as it is generally done manually and is not easily validated, so if there’s a way to collect this information automatically, then we can improve the ability to detect changes more effectively and efficiently.
Telehealth: allows for remote monitoring, improved communication and accessibility to clinicians.
Expediting the Science has to do with more effective and efficient product development. The traditional method of developing drugs and studying its effects on patients is expensive, time consuming and slow. Digital solutions will be able to accelerate the process.
Areas that will bring value to the science of pharma:
- Machine learning: lot of research looks at therapies matching targets and side effect profiles. Slowly getting there, but being able to do it right from the start.
- Genome, proteome: manage data coming in from the field
- Wearables: collect vital signs, certain lab tests, using “exhaust” data from cell phones to infer behavior
- Home monitoring: sleep, activities, reporting of outcomes. Make clinical trial data more robust and data usable
- Diagnostics: identify and track biomarkers
Financing for pharma will change with the move towards a value-based model. Moving away from fee-for-service will result in new reimbursement schema tied to outcomes.
- Machine Learning: for marketing and population segmentation purposes. Targeting assets (patients for medications, areas where there’s not enough support to get molecule to market)
- Big data: leveraging the combination of traditional clinical data with other non-clinical datasets and patient reported data will enable new reimbursable outcomes
- Managing risk: moving to a value-based reimbursement model. Right now fee-for-service, pay for products. In value-based future, pharma will likely take on some of the risk with indication/outcome-based pricing for drugs. Very cumbersome to do that now (i.e. collecting data from different EMRs), but will get easier with new tech and/or new risk bearing contracts.
- Organizational agility: risk-aversive organizations and culture. Current business model for fee-for-service isn’t going to last very long as we continue to move to down the personalized medicine path. Target populations for drug therapy will be more selective and in general decrease. Drug development costs won’t be decreasing as fast. R&D costs amortized a onto smaller population will lead to higher prices.
- Digital Culture: iterations should become cheaper and faster. And pharma may adopt a digital culture that says it’s okay to fail quickly, learn and iterate. Risk will be embraced and not feared.
Pharma is at a juxtaposition with stakeholders in the ecosystem right now because the business model depends on patients who need the drugs. There is no incentive to completely cure people as they will stop being customers. With the current fee-for-service model, pharma doesn’t/can’t benefit from disease cures, so from a financial standpoint, it wants people to be sick enough to still need the drugs. It’s a cyclical business cycle that benefits from people being sick and staying stick.
With a value-based system, pharma will go from the position of “I hope you get sick” to “I hope you get better or don’t get the disease”. Instead of fee-for-service where drugs are a revenue generator, a value-based pharma will naturally shift towards a focus on outcomes and population health, as drugs become a cost center.
This is the same as providers who are now in value-based care arrangements: I make money if you’re healthy. As providers have developed into accountable care organizations, one path forward is that pharma will develop into accountable treatment organizations (taking responsibility for shared risk in therapeutics for its patient).
The value-based pharma model will likely change from drugs as a revenue generator to drugs as an investment/cost-center. For example, being prepaid for a drug budget to make sure the population doesn’t get breast cancer. Providers will want to follow guidelines and ensure that drugs are appropriate for the patient population and the healthcare system will take on a preventative and holistic approach to health.
New Pharma Hires
- Fundamentally pharma isn’t a technology company so will need many different folks to help develop digital health solutions.
- Behavioral specialists important for understanding if patient are responding appropriately because of the drugs.
- Learning specialist: how to get this to population on all sides?
- IoT – patients and doctors can use things that are ubiquitous.
- Managed Care: understanding how pharma will interact with the managed care.
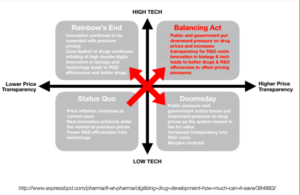
Wayne shares a quadrant analysis by Express Pharma about the 4 scenarios where pharma can go in the future:
Scenario #1: Status Quo (Low Tech, Low Price Transparency)
Pharma is the only industry where a drug that was introduced 10 years ago actually costs more today. Most consumer products drop in price with time and scale.
If nothing changes, low price transparency and price inflation will continue, and any new products entering the market will be at premium prices. However, due to increasing public and political scrutiny, prices will be raised less aggressively than before. Technology enhances innovation but drives very few R&D efficiencies.
Scenario #2: Doomsday (Low Tech, High Price Transparency)
This is the worse case scenario for pharma. The industry reaches a tipping point where public pressure and government action will put downward pressure on drug prices and increase price transparency (high regulation) as the shift from fee-for-service to value-based care continues.
Technology contributes to medical cures just like it has over the last few years, but pharma will fail to leverage technology to generate new efficiencies or drive a step change in cures.
Scenario #3: Rainbow’s End (High Tech, Low Price Transparency)
This is an ideal scenario for pharma. Free marketing pricing and low price transparency continues, and new treatments are priced high. Innovations decrease R&D costs, failures and time to market, leading to better efficiencies. Pharma uses technology to enhance patient adherence, drug targeting, and drives up volumes and durations of use.
Scenario #4: Balancing Act (High Tech, High Price Transparency)
This is the scenario that’s best for all stakeholders.
Pharma innovation using digital technology to balance out pricing and move into new business model. Value-based pricing gains increasing acceptance and technology reduces development time and costs while enabling personalized medicine that delivers the right treatment to the right patient at the right cost.
So there you have it. The story of mPharma.
How will this play out? Will pharma stay in its traditional role or evolve into something completely different? Only time will tell. Keep your eyes open, there may be a surprise ending coming.
Speaker #3
Yan Chow, MD, MBA, is the Medical Director of Digital Health in the Medical Sciences group at Amgen, the world’s largest independent biotechnology company, where he studies the use of wearables, devices, mobile technology, and analytics in clinical trials and programs. He has founded and advised various healthcare startups and holds three U.S. patents.
Current Healthcare Trends
- Patients are empowered by the internet to be consumers. They don’t want information pushed to them. They choose the information they want, and many will trade personal information for a free service.
- Providers share similar sentiments as patients with regards to the internet, and use social media for work. A shift to a value-based model has moved the focus from episodic care to continuous care and population management. This is very different from the ways doctors used to operate.
- Payers believe in real-world evidence and positive socioeconomic impact. They want to know how soon the patient can expect therapies to work, and quantify their ROI. Questions that they’re asking: are patients feeling better? Are they feeling more productive?
Life Sciences
Patients value functional ability (practicality) more than efficacy. The minimum package is not drugs but drugs and a digital solution.
And the FDA is starting to take patient feedback into drug approval process, which has never been done before. The 21st Century Cures Act, which was designed to help accelerate medical product development and speed new innovations to patients faster and more efficiently, requires the FDA to consider non-clinical data.
Back in the day, clinical trials and data were the main determinant of the legitimacy of a drug. But now, real-world evidence and ramifications of the drug (what does it do to the patient?) are emerging as deciding factors in drug development.
Digital Health
There is a low barrier to experimentation in digital health compared to pharma. The freedom to try different things opens up the floodgates, and startups have come in from domains that had never intersected with healthcare before, like robotics, VR/AR, 3-D printing, and social media.
Many different players from non-health industries are jumping into healthcare because that’s where all the money is. With the influx of capital, interest and attention on digital health, we can expect major changes to come.
How is Biopharma Evolving?
- 10 medicines enter Phase 1, one medicine exits at the end.
- Traditional model is very slow, costly, risky. Clinical trials cost $30-40m pre-approval and another $30-40m post-approval.
- Cost of a patient in clinical trial is $10,000.
- Cost of a patient in a digital trial is about $1,000.
- New drug pipelines are dying up. Inability to precisely target right patients in the real world.
New Business Model
- Wants to provide a more holistic, more complete and safer solutions to patients.
- Faster drug development and efficient process. Demonstrate value by having data. Create digital health ecosystem around the medicine and create virtual digital trials at potentially 1/10th of the cost of traditional trials.
- Develop more precise patient targeting.
- Patient recruitment & retention.
Digital Health Technologies

Digital health tech isn’t just mobile apps. Goes from telemedicine (been around for 30 years) to bionics.
Pharma & Digital Health
- Top 12 pharma companies released 700 apps in 5 years. That’s more than an app for every medicine.
- Apps created around social media to create communities around medicine for feedback and optimization.
- Clinical trial streamlining, recruiting, engagement and adherence (Science 37). Providing services to patients at their homes, relieve burden of subject having to go onsite.
- M&A, digital health investments 28/325 deals between 2014 – 2016 were digital health.
- New business models (data driven outcomes-based pricing, risk sharing, open sourcing).
- Accelerators and startups networks and ecosystem (JLABS, Janssen, Amgen)
- Crowdsourcing and open innovation (Innocentive, Indiegogo). Innocentive poses a question to the world that your company can’t solve. You get a prize for solving the problem. The view is that the world has answers you don’t have.
- Nontraditional alliances (Verily, Apple, Qualcomm, IBM Watson. Very data-driven which is very different than how pharma traditionally thinks of data)
- Manufacturing (i.e. 3-D printed pills for epilepsy). Can we print pills that combine medicines into one pill?
Big Takeaway
It’s still a very early time for digital health in pharma right now, changes are coming and things are beginning to move and transform along with everything else in healthcare. There’s a lot of potential and unanswered questions.
We don’t know where future is going, but it will not look like the present because the current situation is not sustainable.